 Remember the periodic trend of the periodic table i.e. Therefore, argon has an atomic radius less than chlorine’s atomic radius. This will mean that adding a proton and electron will decrease the atomic radius.Īs argon is to the right of the chlorine and on the same period. As the proton has higher attraction than the electron, the proton will win over the electron –electron repulsion between the outer electron shell and other shell, it will shrink the outer shell. When we go left to right, we add an electron and proton. This happens because no shells are being added since all the electrons in that period are present in the same shell. The electrons of the elements at the bottom are farther away from the nucleus.Īlso, when we go left to right on the periodic table, the atomic radius will decrease. The atomic radius will increase because the elements at the bottom will have more energy level than the elements above it. When we go down the group (the columns) of the periodic table. We know that relative atomic radius is mostly determined by how many electron shells are there and by the number of protons that are in a nucleus. Let us completely understand the explanation why the atomic radius of argon is less than that of chlorine.Īccording to the periodic table, chlorine is located in group $ 17 $ whereas argon is located in group $ 18 $. We know that there is greater attraction from the nucleus in case of argon because as the electrons increase, effective nuclear charge also increases. First, we should know about the location of chlorine and argon in the periodic table. It does not store any personal data.Hint: In order to answer this question, we should know about the effective nuclear charge of the chlorine and argon atoms. The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. The cookie is used to store the user consent for the cookies in the category "Performance". This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. The cookies is used to store the user consent for the cookies in the category "Necessary". The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". The cookie is used to store the user consent for the cookies in the category "Analytics". These cookies ensure basic functionalities and security features of the website, anonymously. Necessary cookies are absolutely essential for the website to function properly. It makes the radius of Cl⁻ bigger than Cl. There is repulsive force between electrons. 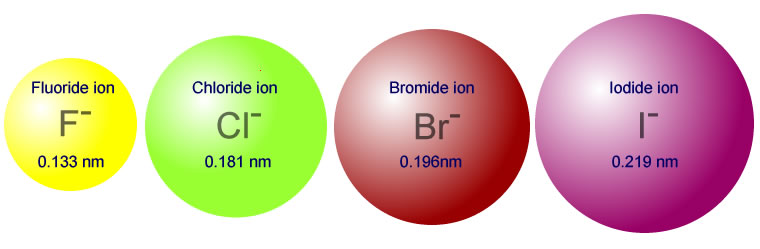
The number of electrons in chloride is 17 electrons. In addition, Cl− ions are larger than Cl atoms because each ion has one more electron, which means greater electron-electron repulsion and an expanded cloud of electrons around the ion’s nucleus. However, Na+ ions are much smaller than Na atoms because their n=3 shell is empty. Why are chlorine ions larger than sodium ions? That will tend to pull the electrons more and more towards the centre of the ion – causing the ionic radii to fall…. 
However, the number of protons in the nucleus of the ions is increasing. Hence, Chlorine will have smaller atomic radius than Sodium.Ĭhlorine/Van der Waals radius Why is the ionic radius of a chloride ion? So, Chlorine’s nucleus size is greater than Sodium. Chlorine has more number of protons than that of Sodium. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
